

The Technology and Style of Executive Materials(2nd ed.).

Volume occupied by atoms hcp how to#
See how to calculate the number of atoms in HCP unit cell In HCP unit cell 1/12 atoms are present in each corner.
Volume occupied by atoms hcp full#
For a full article explaining APF, check out this link. Packing efficiency is defined as the total volume occupied or covered by the spherical shaped particles. APF is the APF is basically the fraction of atoms to void.
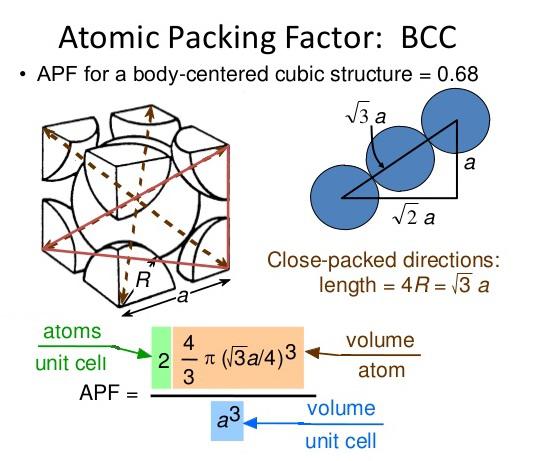

Since we use the hard sphere model, each point inside the cell is either part of an atom, or part of the void. Teaching General Chemistry: A Materials Science Partner(3rd ed.). The Atomic Packing Factor (APF) is essentially the density of the unit cell. displaystyle beginalignedmathrm APF amp =frac Nmathrm atoms Vmathrm atom Vmathrm unitcell =frac 6cappear in frac 43pi l^3frac 3sqrt 32a^2c10ptamp =frac 6cdot frac 43pi ur^3frac 3sqrt 32(2r)^2sqrt frac 23cappear in 4r=frac 6cdepartment of transportation frac 43pi l^3frac 3sqrt 32sqrt frac 23cdepartment of transportation 16r^310ptamp =frac pi sqrt 18=frac pi 3sqrt 2approx 0.740,480,48.endaligned See furthermore edit Recommendations edit After reaching a yield stress at t x 180, t y 110 ( reduced units ), we observe a sudden increase of the amount of hcp structure as demonstrated in Fig. (volume of spheres within the unit cell) / (volume of cell). It can be then probable to compute the APF as follows: A P N = In a capital t o michael beds V a capital t o michael Sixth is v u n i testosterone levels chemical age m l = 6 ⋅ 4 3 π l 3 3 3 2 a 2 chemical = 6 ⋅ 4 3 π l 3 3 3 2 ( 2 l ) 2 2 3 ⋅ 4 r = 6 ⋅ 4 3 π r 3 3 3 2 2 3 ⋅ 16 ur 3 = π 18 = π 3 2 ≈ 0.740 480 48. atomic coordination geometry hexagonal but interstitial coordination geometry is. So the volume of the hcp unit cell transforms out to end up being (3/2) √ 3 a 2 ddisplaystyle a^2c, that is usually 24 √ 2 r 3displaystyle ur^3. If a = 2 rdisplaystyle a=2r, then its elevation can end up being easily determined to end up being 8 3 adisplaystyle sqrt tfrac 83a, and, consequently, c = 4 2 3 urdisplaystyle d=4sqrt tfrac 23r. Obviously, the edge of this tetrahedron is certainly adisplaystyle a. y., twice the elevation of the regular tetrahedron whose vertices are usually occupied by (state) the central atom of the lower level, two adjacent non-central atoms of the same layer, and one atom of the center layer 'resting' on the prior three. The last mentioned is double the length between surrounding levels, i. Now let adisplaystyle abe the side length of the bottom of the prism and ddisplaystyle dbe its height. So the total quantity of atoms in the cell will be 3 + (1/2)×2 + (1/6)×6×2 = 6. Certainly, three are the atoms in the center level (inside the prism) in add-on, for the top and bottom level layers (on the facets of the prism), the central atom will be propagated with the surrounding mobile, and each of the six atoms at the vertices will be shared with various other five surrounding tissues. We find, within error, a similar atomic volume, VA 8.274 (7) 3 for the fcc structure and VA 8.269 (11) 3 for the hcp structure and a large coexistence. Here the unit cell (equivalent to 3 ancient unit tissues) will be a hexagonal prism filled with six atoms (if the particles in the crystal are usually atoms). With this work, we also demonstrate the possibility of routine static high-pressure experiments with conventional bevelled diamond-anvil geometry in the 0.3–0.4 TPa regime.For the hexagonal close-packed structure the derivation is definitely similar. Here we confirm this observation with a synchrotron-based X-ray diffraction experiment carried out within a diamond-anvil cell and report indications of the onset of the transition towards a bcc structure at pressures beyond 320 GPa. In atomic systems, by convention, the APF is determined by assuming that atoms are rigid spheres. It is a dimensionless quantity and always less than unity. The hcp phase was identified at 217 GPa in an experiment (Akahama et al., 2006), and the bcc phase has been recently confirmed in a dynamic ramp-compression experiment coupled with time-resolved X-ray diffraction (Polsin et al. In crystallography, atomic packing factor (APF), packing efficiency, or packing fraction is the fraction of volume in a crystal structure that is occupied by constituent particles. For aluminium, first principle calculations have consistently predicted a phase transition sequence fcc–hcp–bcc in a pressure range below 0.5 TPa. At extreme pressures, common materials are thus expected to transform into new dense phases with extremely compact atomic arrangements that may also have unusual physical properties. With the application of pressure, a material decreases in volume as described in its equation of state, which is governed by energy considerations.

